Innovative Health, a single-use cardiology medical device reprocessing company, has announced FDA clearance for reprocessing the Webster Duo-Decapolar Diagnostic EP Catheter. The 20-electrode EP catheter is used in electrophysiological mapping of cardiac structures, such as in Atrial Fibrillation procedures.
Innovative Health already has clearance to reprocess a large portfolio of Biosense Webster’s diagnostic, ultrasound, and mapping catheters, including the PENTARAY Nav eco High-Density mapping catheter, which was cleared just a few days earlier. The company says the Webster Duo-Decapolar Diagnostic EP Catheter costs $1,000 new and represents another important means for EP labs to significantly reduce procedure costs. “Many EP labs across the country rely on us to reduce procedure costs,” said Innovative Health CEO Rick Ferreira in the statement. “As a specialty reprocessor, Innovative Health is able to focus on a specific clinical area, develop deep clinical and scientific competencies, and work with FDA on our submissions to achieve clearance and make higher savings available.”
Unlike reprocessors of reusable devices, reprocessors of single-use medical devices must achieve FDA clearance to market medical devices and are under strict regulatory oversight, says Innovative Health, pointing out that single-use device reprocessing is used by 19 of the top 20 US News & World Report hospitals. However, not everyone is supportive of the process, including a Healthcare Purchasing News social media reader who commented on a separate story: “Nope. Not worth the risk. This is a step in the wrong direction.” HPN caught up with Innovative Health's Lars Thording, VP, Marketing & Public Affairs to get the company's take on the controversy.
“There is not much resistance among clinicians anymore to the use of reprocessed catheters,” Thording asserted. “It has been established that these are as safe and effective as new ones, and FDA data shows that reprocessed catheters do not fail more frequently than new ones. At the same time, as electrophysiology devices have become more advanced, specialized reprocessors have evolved their capabilities and are able to demonstrate to FDA that they can indeed clean and test the devices. Our hospital partners continue to ask these OEM representatives for data to support the notion that reprocessed devices are not equivalent to new ones, but no such data has been presented. Ultimately, FDA’s clearance process establishes that reprocessed devices are substantially equivalent to new ones and don’t present added risk to the patient.”
SUD evolution
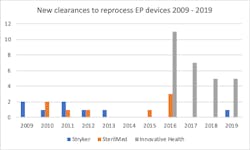
"The market for and utilization of reprocessed single-use devices has been changing over the last few years," Thording told HPN. "In the late 2000s, the market was dominated by two large reprocessors who sought to reprocess everything that could be reprocessed – from compression sleeves over laparoscopic scalpels and trocars to electrophysiology (EP) devices. Hospitals were experiencing massive savings from these programs. Between 2009 and 2011, these reprocessing companies were acquired by large manufacturers – Stryker (Ascent Healthcare Solutions) and Johnson & Johnson (SterilMed). Since then, the industry has suffered from a deterioration in the efficiency of these programs, because of the service level and because of technologies being removed from the market: The old FDA clearances that reprocessors owned did not cover new devices.Specifically, the number of clearances for new devices from established, broad reprocessors has gone down, resulting in a challenge for hospitals that need their reprocessing savings to continue to make an impact on their costs,"Thording continued. "Cost savings from reprocessing can only grow if reprocessors continue to get clearances for new technologies – otherwise savings die when technologies are discontinued. In the electrophysiology space, technology development is very fast – and technologies are obsoleted only after a few years in the market. Given the lack of new clearances, this means that savings have gone down. But this has also lead to the emergence of a new type of reprocessor: The specialty reprocessor, less focused on wholesale discount sale of new products, and more focused on deep clinical and scientific commitment to a therapy area.
The specialty reprocessor – like Innovative Health - is an inch wide and mile deep – as opposed to trying to cover the whole spectrum of procedures. As a result of this, FDA clearances for new and more complex technologies are possible. To increase savings in electrophysiology – Innovative Health’s area of focus – new technologies must be embraced, and given the impact electrophysiology has on heart diseases these years, manufacturers are aggressively developing new products. The reprocessor is always two steps behind, and only a strategic investment in regulatory and scientific competencies can keep up with new technology development.
Innovative Health has received 28 EP clearances since 2016, 5 in 2019 alone. Some of these are for technologies that have previously not been considered reprocessable. The recent PENTARAY clearance is an example of this, and the addition less than a week later of the Duo-Decapolar diagnostic catheter provides evidence to the same commitment: Constant new clearances that help hospitals sustain and grown their reprocessing savings in the EP lab, so that hospitals can provide the important procedures to more patients.